Deep Learning Pipeline for Microscopy Denoising and Registration
A self-adaptive deep learning framework integrating DeepCAD denoising and PyLoReg registration. Designed for high-fidelity restoration of in vivo calcium imaging by eliminating both Poisson noise and complex motion artifacts.
Demo Applications
Continuous 24-hour High-Resolution Intravital Imaging in Freely Behaving Mice
This video demonstrates continuous 24-hour calcium imaging of the medial prefrontal cortex (mPFC) in a freely behaving mouse, acquired via miniature two-photon microscopy at an imaging depth of 1800 µm. The video synchronizes a 24-hour circular ethogram with concurrent behavioral and neural data. Top panels display infrared behavior alongside Raw and NeuroPilot-processed calcium imaging. Extracted neuronal footprints and a normalized calcium heatmap are temporally aligned with the color-coded behavioral timeline.
Title
This video demonstrates the longitudinal performance of NeuroPilot in segmenting individual spinal neurons and extracting calcium traces from free-moving mice across 28 days (Days 1, 5, 14, 21, and 28). Left: STD projection map overlaid with color-coded spatial masks for individual neuron identification. Middle and Right: Synchronized comparison of temporal calcium traces extracted from raw data (white) and NeuroPilot-processed data (green).
NeuroPilot-Driven Signal Recovery of Head-mounted Three-photon Calcium Imaging across Cortical and Hippocampal Depths
This volumetric video demonstrates the spatiotemporal restoration of low-signal-to-noise ratio (SNR) data acquired via miniature three-photon microscopy. Left: The raw 3D rendering exhibits significant noise and signal attenuation, particularly in deeper tissue layers. Right: The corresponding volume processed by NeuroPilot.
Enhanced Large-scale Two-Photon Calcium Imaging of the Mouse Cortex via NeuroPilot
NeuroPilot-enhanced large-scale cortical two-photon imaging. Real-time comparison between raw two-photon recordings and NeuroPilot-processed results. The video demonstrates signal restoration across a wide-field cortical area (encompassing a ~6 mm field of view) and within magnified local regions (regions 1-3).
Image Enhancement for Multiplexed Two-photon vS1 Imaging
This video demonstrates the denoising and restoration capabilities of NeuroPilot on in vivo multiplexed two-photon microscopy data from mouse vibrissal primary somatosensory cortex (vS1, barrel cortex) neurons. The imaging captures both calcium dynamics (GCaMP6s, green) and structural morphology (mCherry, magenta TODO).
NeuroPilot Performance on Spinal Cord Vascular Imaging
High-fidelity restoration of spinal microcirculation in freely moving mice via NeuroPilot. This video demonstrates NeuroPilot’s performance in restoring two-photon imaging data of the spinal cord vasculature from a freely moving mouse.
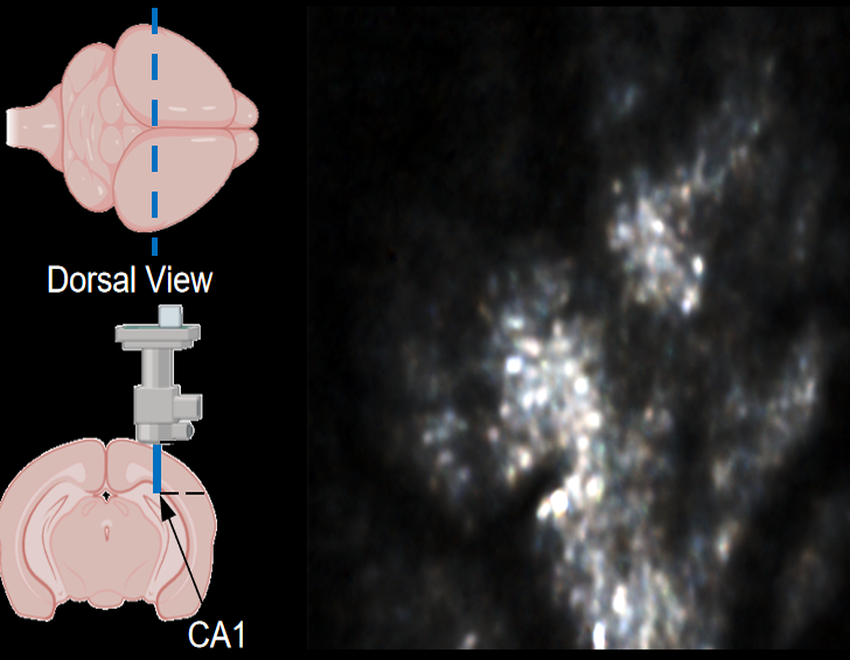
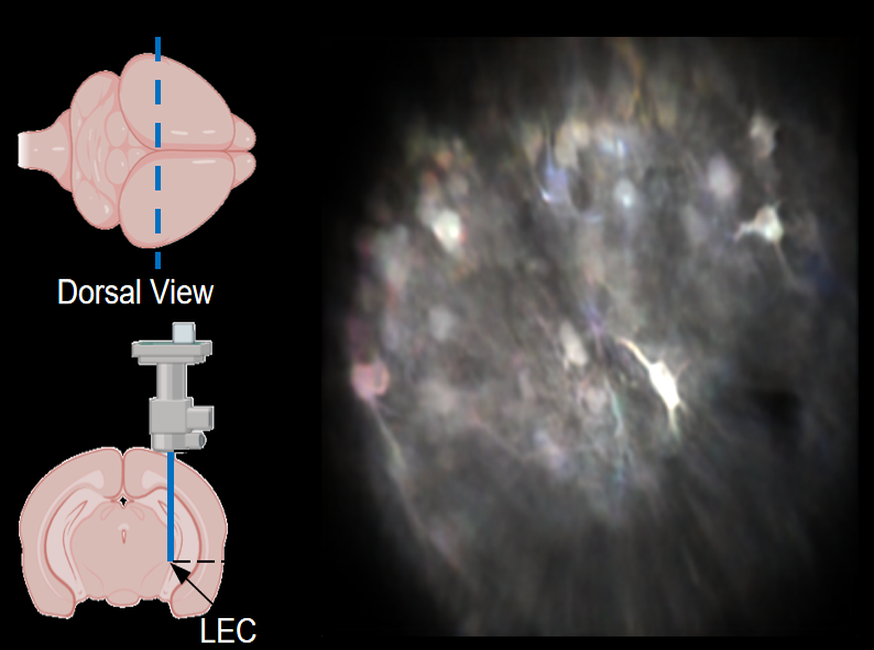
Robust Motion Correction and Single Restoration in LEC and CA1
This video demonstrates stable, high-fidelity calcium imaging in two deep-brain regions of interest (ROIs) within mice, achieved through miniature two-photon microscopy and processed via NeuroPilot.Target Region 1: Lateral Entorhinal Cortex (LEC). Microendoscopy (via a GRIN lens) provided access to LEC neurons at a physical depth of 4.3 mm. Target Region 2: CA1 Regions. Imaging was performed at a physical depth of 1200 µm.
Mitochondrial Calcium Signal Recovery and Morphology Restoration
This video demonstrates the restoration of mitochondrial calcium dynamics and network topology from noisy two-photon microscopy data using NeuroPilot. Left: Fluorescence images comparing raw data (top) and NeuroPilot outputs (bottom). Insets highlight the recovery of continuous tubular structures. Right: Skeletonization analysis demonstrating that NeuroPilot accurately extracts the true mitochondrial network from noise.
Stable Cross-Session Tracking of Spinal Neurons During Varied Stimulation
This video demonstrates the stability of single-cell tracking in the mouse spinal cord across various external stimulations. Left: Illustrations showing physiological states, including free-moving, ice stimulations, mechanical stimulations, and spinal fixation. Middle and Right: Synchronized raw and NeuroPilot-processed recordings. Kymographs (bottom) demonstrate the superior temporal and spatial stability achieved after processing.
Longitudinal Segmentation and Signal Extraction of Spinal Neuronal Activity over 28 Days
wait
Longitudinal Tracking of Mouse Spinal Cord Neurons over 28 Days
This video demonstrates in vivo longitudinal imaging of spinal cord neurons in an SNI (spared nerve injury) model. Data was acquired via miniature two-photon microscopy over five recording sessions (Days 1, 5, 14, 21, and 28).Top row: Raw recordings showing progressive noise and signal degradation. Bottom row:NeuroPilot-processed outputs.